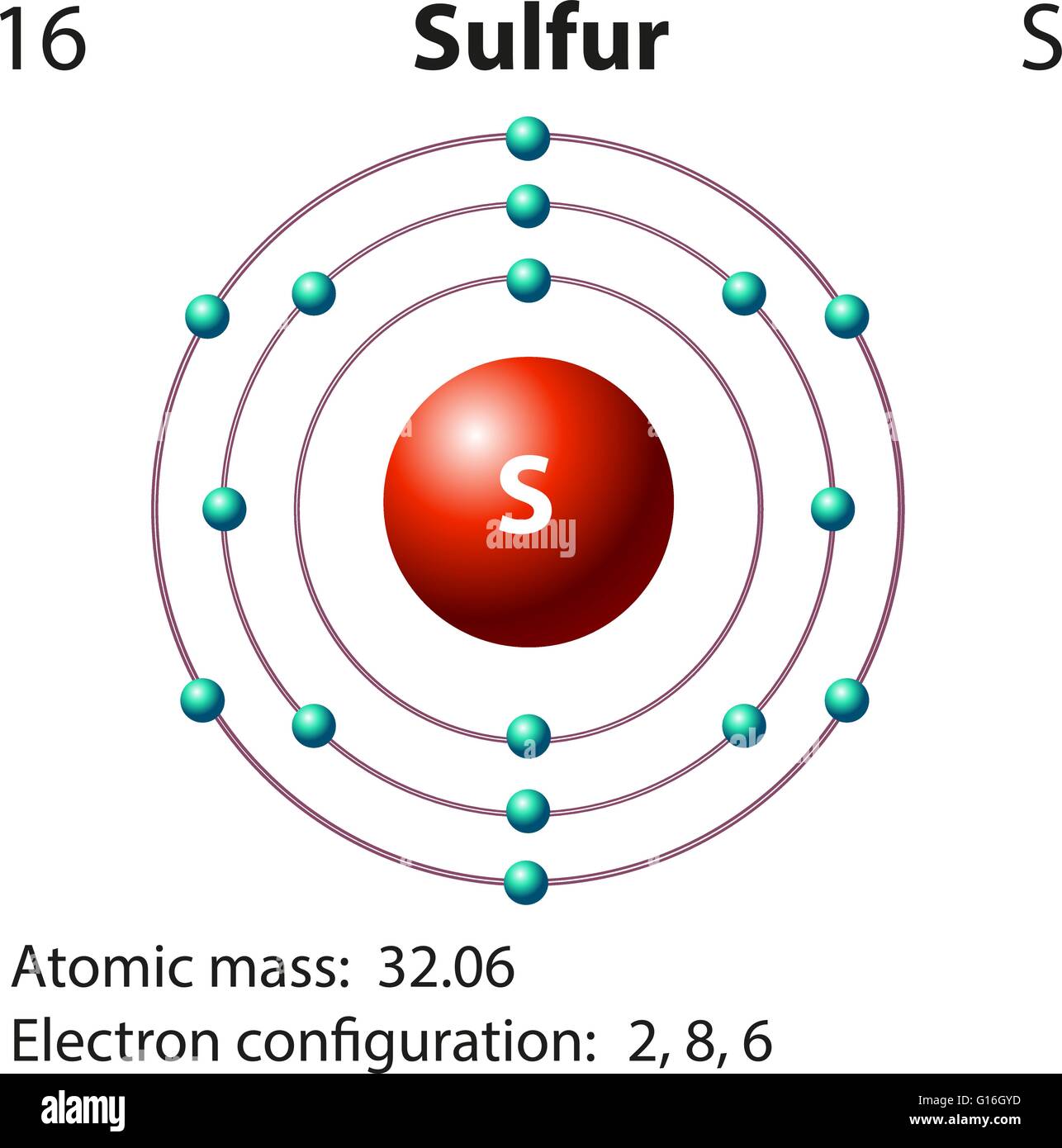
The number of electrons in an atom equals that atom’s atomic mass. Let us first calculate the number of electrons in the sulfur atom. In the next step, we will draw the shells containing electrons in the sulfur atom. Thus, the Sulfur atom consists of 16 protons and 16 neutrons. Here, p+ represents protons while n° represents neutrons. Therefore, the nucleus of the Sulfur atom appears as drawn above. Now, we will draw the nucleus of the Sulfur atom. Hence, the number of neutrons in the Sulfur atom ═ 16 Number of neutrons = Nearest whole number obtained by rounding up atomic mass – Number of protonsĬalculating the number of neutrons in the Sulfur atom ═ 32 ˗ 16 ═ 16 The formula for the number of neutrons is given as: The number of neutrons is obtained by rounding up the atomic mass to the nearest whole number and then subtracting the number of protons out of it. Let us now calculate the number of neutrons in the Sulfur atom. Therefore, the number of protons in the Sulfur atom is also 16. In the case of the Sulfur atom, the atomic number is 16. The atomic number of an atom is equal to the number of protons in that particular atom. We will begin by drawing the nucleus of the Sulfur atom.Īs the nucleus of an atom comprises protons and neutrons we will first calculate the number of these atomic species. We will use this information to draw the Bohr model of Sulfur atom. The electronic configuration of Sulfur is 3s 23p 4.The information that we can infer from the above-mentioned Sulfur box is as follows: Sulfur is the 16 th element of the Periodic table. The electrons are distributed in three shells, viz. In the case of sulfur, there are 16 protons, 16 neutrons, and 16 electrons. These electrons are responsible for the formation of chemical bonds between two or more atoms. The electrons located in the highest energy level of an atom are known as valence electrons while the shell is known as the valence shell. They move from lower to higher energy levels when they absorb energy, while they fall from higher to lower energy levels as they lose energy. The electrons are also capable of shifting their orbits. The energy of electrons increases as the distance between the electron and the nucleus increases. The electrons nearest to the nucleus have the least energy. The negatively charged electrons are the movable species that rotate around the nucleus in fixed circular paths. The nucleus is stationary and the maximum mass of the atom is concentrated in it. As per this model, an atom consists of a positively charged nucleus comprising two atomic species, positively charged protons and neutral neutrons. This model came to be known as the Bohr˗Rutherford model or Bohr model of atom. with the lowest number assigned to the innermost shell. while in the numerical order these were named 1, 2, 3,4, etc. In the alphabetical order, these shells were named K, L, M, N, etc. He named these orbitals in ascending order from inside to outside. This is why these orbits were also known as energy levels or shells. Each of these orbits was said to have fixed energy. He explained that the electrons do not travel in a random path but follow definite orbits while moving around the nucleus. Later, in 1915 Niel Bohr modified this model. However, it could not explain the fact how will the atom remain stable as the moving electrons will undergo acceleration and emit energy. The model rightly explained the movement of electrons around the nucleus of the atom. The major one was that the model did not speak about the stability of the atom. Although this model was widely accepted, there were certain drawbacks that could be explained by him. Ernest Rutherford gave the planetary model of the atom in 1911.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
